
by Sharon Beder
Introduction
Human Skin Cancer
Sunscreen and Fabric
The Mouse Model of Cancer
Studies Using Skin Tissue
Drugs and Sunlight
Plant and Algae Growth
Conclusion
Glossary
Bibliography
What Causes Ozone Depletion?
The ozone in the ozone layer is being broken down by chlorine atoms from chlorofluorocarbon (CFC) molecules and bromine atoms from halons. CFCs and halons are produced by humans used in many applications such as refrigerants, anaesthetics, aerosols, fire-fighting equipment and the manufacture of materials such as stryrofoam. They were thought to be completely safe, chemically inert, and environmentally neutral. However it was soon found that they were not so ideal when they reached the upper atmosphere.
When CFCs reach the upper atmosphere they are first degraded by the very high energy of UV (ultra-violet) radiation. Degradation of CFC leaves a free chlorine atom. The basic cause of ozone layer depletion is that this chlorine atom then breaks up ozone molecules. Ozone then disappears. The chlorine atoms are recreated in subsequent reactions. One chlorine atom can destroy 100,000 ozone molecules.
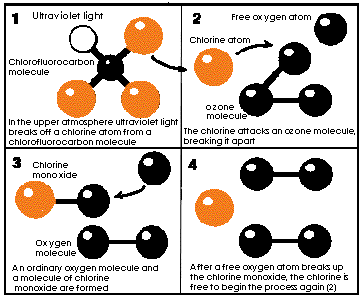 Chlorofluorocarbon molecules in the stratosphere release chlorine atoms which
break up ozone molecules to form ozygen. (diagram adapted from Time Australia
Magazine 19 October 1987)
Chlorofluorocarbon molecules in the stratosphere release chlorine atoms which
break up ozone molecules to form ozygen. (diagram adapted from Time Australia
Magazine 19 October 1987)
Degradation of halons leaves a free bromine atom that destroys ozone in the same way as chlorine. CFCs and halons last a long time (100 years on average). After it was confirmed that an environmentally disastrous side-effect was taking place in the upper atmosphere, plans and agreements were made to phase out CFCs and halons. However, at the moment CFCs and halons are still being produced and are being put into the stratosphere about 5 times as fast as they can be removed naturally.